Description
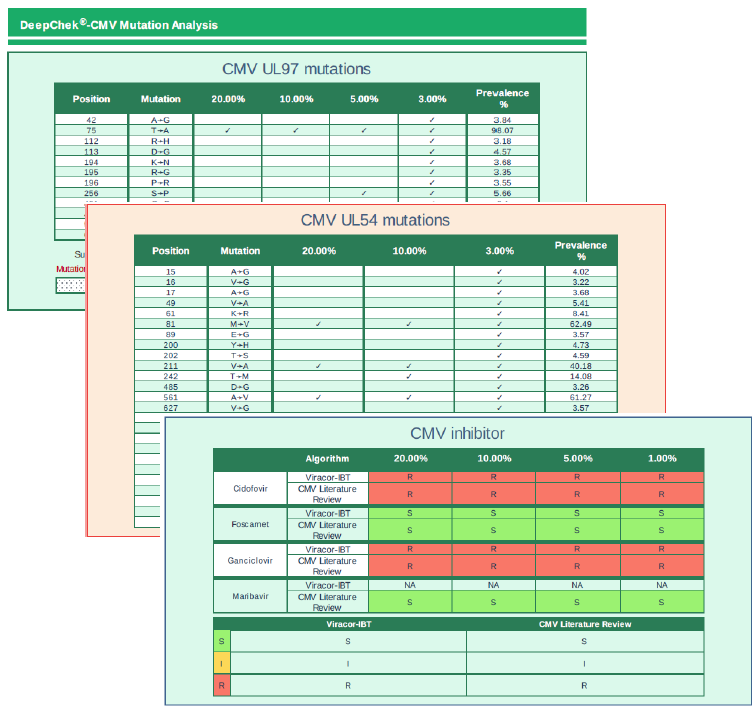
The DeepChek® Assay -CMV UL54/UL97 (RUO) is intended to determine antiviral drug resistance to cidofovir, foscarnet sodium, ganciclovir and maribavir through PCR/sequencing (NGS).
Methodology
DNA Sequencing • Polymerase Chain Reaction (PCR)
More information on the DeepChek® Assays – Click here
More information on the DeepChek® Software – Click here
CHARACTERISTICS AND PERFORMANCES
EXAMPLES OF REPORTS